Alicia O. Hernandez-Castillo Investigating Cyclic Diketone Molecules Using Rotational Spectroscopy
Investigating Cyclic Diketone Molecules Using Rotational Spectroscopy
Diketones are a family of compounds extensively investigated. We have acquired the broadband rotational spectrum of these two molecules using both the spectrometer at UC Davis and the one here at HMC. The spectroscopic data has been analyzed with the aid of ab-initio calculations to yield a complete set of molecular parameters. In the process we have been able to gain insight on the structure of the molecules and compare the subtle differences between the structures.

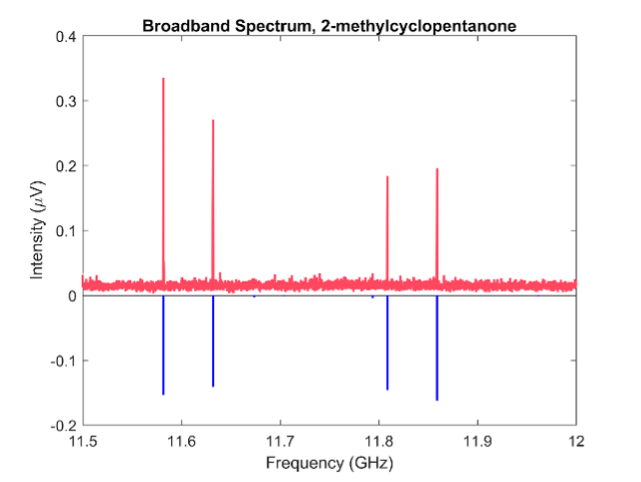
We are in the process of finishing the analysis of 2-methylcyclopentanone and 2,2-dimethylcyclopentanone where we have recorded spectra in the 26.5-40 GHz frequency region where we can observe 13C in natural abundance. Moreover, we have used these molecules to optimize the HMC CP-FTMW spectrometer to get better signal to noise.